15th November 2018
A number of countries – including Switzerland, the UK, Germany, the Netherlands, and Canada – prescribe heroin for use under medical supervision, as part of successful programmes to treat long-term users of illicit opioids. Recently Glasgow and Durham, UK, have put forward proposals for Heroin-assisted treatment (HAT) aimed to combat public health risks caused by street use.
HAT is significantly less common than opioid substitution treatment (OST). This is because HAT is typically reserved for opioid users who have proven unresponsive to other forms of treatment, and because it is considered more politically controversial. But despite its relatively limited availability, there is now a substantial body of evidence demonstrating the effectiveness of HAT. This evidence provides useful lessons for managing one of the most risky and problematic forms of drug use as a public health challenge, rather than a criminal justice one.
Legally recognised as a medical intervention and therefore not prohibited by the UN drug conventions, HAT shows the wider potential benefits – locally, nationally and internationally – of moving the supply and use of an illicit drug into a completely legal, strictly regulated market. The experience of Switzerland, which became a pioneer of HAT in the 1990s, represents the most comprehensive source of empirical evidence on the outcomes of such a transition. It suggests that if this form of treatment were rolled out widely – particularly in major consumer countries – it could have major benefits for many people dependent on heroin, and a significant impact on the scale of illicit drug markets.
Background

The prescribing of medical-grade heroin as a treatment for heroin dependence has a long history, having been firmly established in UK medical practice by the 1926 Rolleston Committee,1 after which it operated in parallel with the criminalisation of non-prescribed heroin under both domestic and international law.2 Coming to be known as the ‘British system’, it remained in place until concerns around rising heroin use among young people, overprescribing, and the risk of the drug being diverted to the illicit market, led to heavy restrictions being introduced in 1967. Despite an exponential rise in use since then, today less than 200 of the UK’s more than 200,000 users receive heroin on prescription.
Switzerland, like much of Europe, experienced a rapid rise in injecting heroin use during the 1970s and 1980s, but ultimately adopted a very different policy model to the UK. By the 1980s, heroin use had graduated into a full-blown public health crisis, as it became clear that illicit injecting – and particularly high-risk behaviours such as the sharing of needles – was associated with high rates of HIV transmission. In 1986, Switzerland had approximately 500 HIV cases per million people, the highest proportion in Western Europe at the time.3 By 1989, half of all new cases of HIV transmission were linked to illicit drug injection.4 By 1990, HIV prevalence was over 40% among those who reported having used drugs for more than 10 years, and in the era before effective treatments for HIV/AIDS, mortality rates among this population were correspondingly high. Added to this, there were growing fears about sexual contact with injecting drug users leading to a rise in HIV infection rates among the wider population.
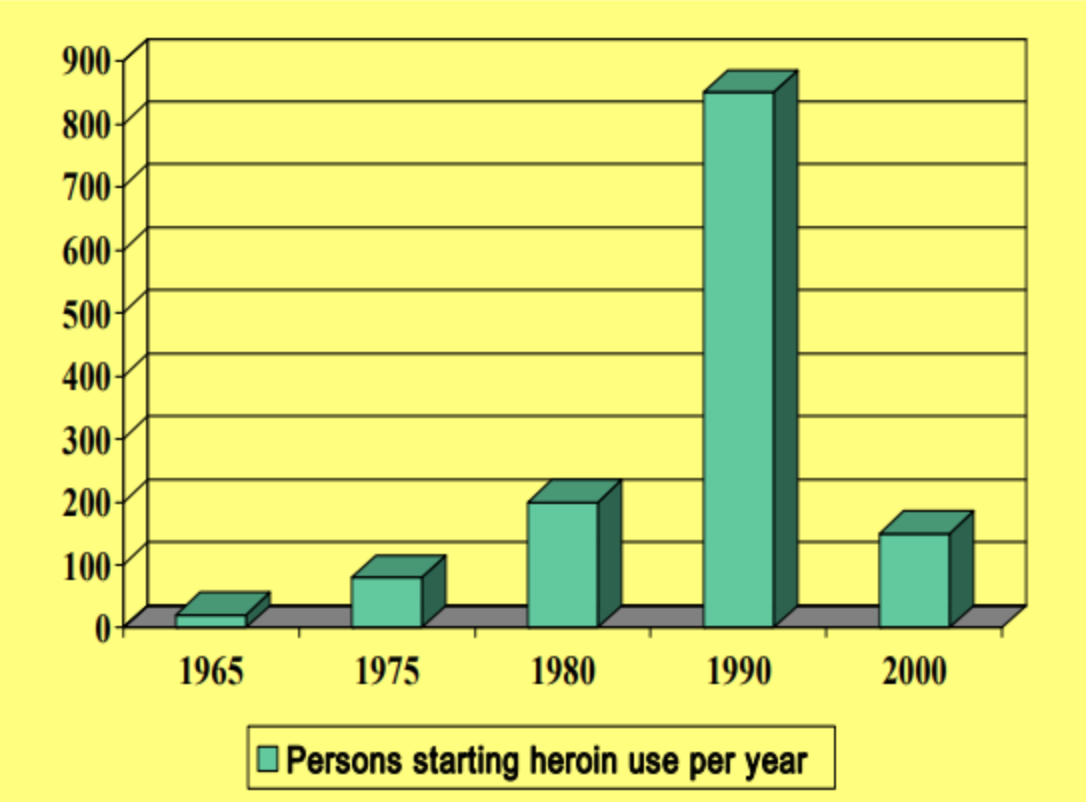
As this challenge grew, initial responses consisted mostly of traditional law enforcement crackdowns. Switzerland’s federal drug law was revised in 1975, to include a greater focus on abstinence, which led to significantly increased arrests, and mandated registration of illicit drug users and sellers by the police – rejecting harm reduction measures such as needle and syringe programmes (NSP), and imposing onerous licensing requirements on methadone prescribing. The response failed, with illicit drug injection and related health problems continuing to increase sharply. Zurich became a particular focus, with the number of people who inject drugs growing from less than 4,000 at the time of the 1975 law revision, to an estimated 10,000 in 1985, 20,000 in 1988, and 30,000 in 1992.5
As Zurich’s street drug scenes became an increasingly visible, problematic and politically charged manifestation of the injecting phenomenon, new approaches were demanded. In 1987, the city authorities made a pragmatic decision, attempting to contain and manage the problem by establishing a tolerance zone – the Platzspitz park – where people were allowed to use drugs. The space soon became known as ‘Needle Park’, and it did enable the injecting scene to be contained and managed to some degree, as well as facilitating the targeted provision of health services. Between 1988 and 1992, the ZIPP-AIDS project based in the park responded to 6,700 overdose episodes, vaccinated thousands for hepatitis B, and distributed 10 million sterile syringes.6
The ongoing health and crime problems linked with needle park, particularly those that spilled into neighbouring areas, ultimately led to its abrupt closure in 1992. In an example of the so-called ‘balloon effect’,7 the drug scene simply shifted elsewhere, and problems continued. However, the way in which the intervention prioritised health over enforcement helped shape the discussions around policy responses to drug injection that followed. Once again, it became clear that new thinking was needed.
In 1991, at the request of municipal authorities and state (canton) governments, a new national programme was established within the Federal Office of Public Health to reconsider the problems. Reflecting previous experiences, the recommendations that emerged were public health-led, including a combination of established harm reduction interventions (OST and NSP), treatment and social support provision, and a new call to explore HAT. In 1992, a change in the law enabled such an exploration.
The Swiss HAT model differed from the old British System in that rather than being given ‘takeaway’ prescriptions, patients were required to attend a clinic once or twice a day and to use their prescriptions on site under medical supervision. The idea was to combine the benefits of prescribed supply (heroin of known strength and purity, free from contaminants and adulterants, and used with clean injecting equipment) with the benefits of regular access to services and supervised use in a safe and hygienic venue (as found in the more common supervised injection facilities such as Canada’s Insite facility8), while also preventing the diversion of prescribed heroin to the illicit market.
The first HAT clinics opened in 1994 as part of a three-year national trial. In late 1997, the federal government approved a large-scale expansion of the trial, aimed at accommodating 15% of the nation’s estimated 30,000 heroin users, specifically those long-term users who had not succeeded with other treatments.
The programmes were explicitly designed and implemented as an empirical investigation. They were rigorously documented and evaluated, and evolved in line with the results generated, following public consultation and debate. In this way, it was possible for the policy model to grow from a scientific experiment into a more formalised policy framework that enjoyed growing public support – a process helped by overwhelmingly positive outcomes.
Summary of Impacts
Changing the law or regulatory infrastructure to allow heroin prescribing, while important, has not driven all the positive outcomes listed below by itself. These outcomes also reflect the wider realignment from a criminal justice to a public health model, and the investment in services that has followed. However, the change in policy and law, much like the introduction of decriminalisation approaches in other countries, has helped facilitate this shift.
- Health outcomes for HAT participants improved significantly
- Heroin dosages stabilised, usually in two or three months, rather than increasing as some had feared
- Illicit heroin (and illicit cocaine) consumption was significantly reduced
- A large reduction in fundraising-related criminal activity among HAT participants. (This benefit alone exceeded the cost of the treatment9)
- Heroin from the trials was not diverted to illicit markets
- Initiation of new heroin use fell (the medicalisation of heroin making it less attractive), and, in turn, there were reductions in street dealing and recruitment by ‘user-dealers’10 11
- Uptake of treatments other than HAT, especially methadone, increased rather than declined (as some had feared it might)
These positive outcomes have been reproduced in other countries that employ the Swiss-style HAT model. A 2012 review of these programmes by the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) concluded that HAT treatment can lead to:12 ‘the “substantially improved” health and well-being of [participants]; “major reductions” in their continued use of illicit heroin; “major disengagement from criminal activities”, such as acquisitive crime to fund their drug use, and “marked improvements in social functioning” (e.g. stable housing, higher employment rate).’13 A 2011 review from the renowned Cochrane Collaboration – which is widely seen as providing ‘gold-standard’ reviews of healthcare evidence – came to similar conclusions.14
Despite this evidence of effectiveness, there is limited availability of HAT even in the relatively small number of countries where it exists; it is only available under strict criteria, including long-term use and failure to respond to other treatment programmes. It is possible that the benefits of HAT could be extended if the barriers to access were lower. So far there has only been one study into this possibility, which found that, compared to OST, HAT produced no difference in health outcomes but did produce far greater reductions in illicit drug use.15
Other treatment models similar to HAT, such as prescribing smokable heroin, heroin ‘reefers’, or smokable opium have been tried but, so far, inadequately researched. The HAT model could also potentially be adapted for other currently illicit drugs. Indeed, there are already drug-of-choice prescribing programmes for dependent users of amphetamines.16
Cost-effectiveness
There have also been studies considering the cost-effectiveness of HAT in three countries – Switzerland, Germany and the Netherlands. These report the costs of such programmes to be between €12,700 and €20,400 per patient per year – considerably higher than the cost of OST (with methadone estimated at between €1,600 and €3,500 per patient per year). This is due to both the greater costs of the drug and the additional costs of establishing and maintaining supervised facilities. But the studies also show that, despite the higher cost, the expenditure is more than compensated by ‘significant savings to society’, including less spent on criminal procedures and imprisonment. The EMCDDA notes that ‘If an analysis of cost utility takes into account all relevant parameters, especially related to criminal behaviour, [HAT] saves money’.17The agency concludes: ‘While [HAT] may be a useful addition to our treatment “toolbox” for opioid users, it is not a solution for the heroin problem … But for those among whom the benefit is observed, there are major gains for themselves, their families and society.’
In Switzerland, the generally successful outcomes are also reflected in shifting public opinion. Three-quarters of the population identified drugs as one of the five major problems facing the nation in the mid-1990s; that figure had fallen to one eighth by 2007.18 Reducing highly visible, public drug use was certainly identified as a key driver of support for HAT.19In a national referendum in 2008, the Swiss public voted by a resounding margin to make the programme permanent.20
International Benefits
It has been estimated that just the 10% heaviest users of heroin in Switzerland (most of whom fall into the HAT target group) consume around 50% of all the heroin imported.21 As a result, the reduction in their consumption of illicit drugs as they enter the HAT programme (and the absence of any increase in new heroin users) could lead to a substantial decline in the overall production and transit of illicit heroin for use in the country. So in addition to the potential benefits on an individual and domestic level for consumer nations, if these programmes were rolled out widely, it could significantly reduce the global demand for illicit heroin. This in turn would lead to a corresponding reduction in illicit production, transit and supply – and the vast criminal costs they generate.
References
- British Medical Association Board of Science (2013) ‘Chapter 5 – Drug policy in the UK: from the 19th century to the present day’ in Drugs of dependence: the role of medical professionals.
- The relevant legislation being the UK Dangerous Drugs Act of 1920, and the Hague International Opium Convention of 1912.
- European Centre for the Epidemiological Monitoring of AIDS (EuroHIV) (1999) ‘HIV/AIDS surveillance in Europe’, Saint-Maurice, France.
- Savary, J.F., Hallam, C., and Bewley-Taylor, D. (2009) ‘The Swiss four pillars policy: an evolution from local experimentation to federal law (Briefing Paper no. 18)’, The Beckley Foundation.
- Grob, P. J. (2009) ‘Zürcher “needle-park”: ein Stück Drogengeschichte und politik 1968–2008’, Zurich: Chronos Verlag.
- Ibid.
- The balloon effect describes the way in which drug law enforcement, rather than eliminating the drug problem, often merely displaces it to new locations – like air moving around in a squeezed balloon.
- Killias, M. (2009) ‘Commentaires sur Peter Reuter/Dominic Schnoz, “Assessing drug problems and policies in Switzerland, 1998–2007”’.
- Killias, M. and Aebi, M. (2000) ‘The impact of heroin prescription on heroin markets in Switzerland’, Crime Prevention Studies, vol. 11, pp. 83-99.
- Reuter, P. and Schnoz, D. (2009) ‘Assessing drug problems and policies in Switzerland, 1998–2007’, Swiss Federal Office of Public Health.
- European Monitoring Centre for Drugs and Drug Addiction (2012a) ‘New heroin-assisted treatment: Recent evidence and current practices of supervised injectable heroin treatment in Europe and beyond’, Lisbon: Portugal.
- European Monitoring Centre for Drugs and Drug Addiction (2012b) ‘EMCDDA report presents latest evidence on heroin-assisted treatment for hard-to-treat opioid users’, 19.04.12.
- Ferri, M., Davoli, M. and Perucci, C.A. (2011) ‘Heroin maintenance for chronic heroin-dependent individuals’, Cochrane Drugs and Alcohol Group.
- Haasen, C., Verthein, U., Eiroa-Orosa, F.J., et al. (2010) ‘Is heroin-assisted treatment effective for patients with no previous maintenance treatment? Results from a German randomised controlled trial’, European Addiction Research, vol. 16, pp. 124-130.
- Merill, J., et al. (2004) ‘Dexamphetamine Substitution as a Treatment of Amphetamine Dependence: A Two-Centre Randomised Controlled Trial’, UK Department of Health.
- EMCDDA (2012b) ‘EMCDDA report presents latest evidence on heroin-assisted treatment for hard-to-treat opioid users’, 19.04.12.
- Reuter, P. and Schnoz, D. (2009) op. cit.
- Gutzwiller, F. and Uchtenhagen, A. (1997) ‘Heroin Substitution: Part of the Fight Against Drug Dependency’, in Lewis, D., Gear C., Laubli Loud, M., and Langenick-Cartwright, D. (eds.) (1997) The Medical Prescription of Narcotics: Scientific Foundations and Practical Experiences, Seattle: Hogrefe and Huber Publishers.
- Associated Press (2008) ‘Swiss Vote to Keep Program Giving Addicts Heroin’, 30.11.08.
- Killias, M. and Aebi, M. (2000) ‘The impact of heroin prescription on heroin markets in Switzerland’, Crime Prevention Studies, vol. 11, pp. 83-99.
- Ibid.




